 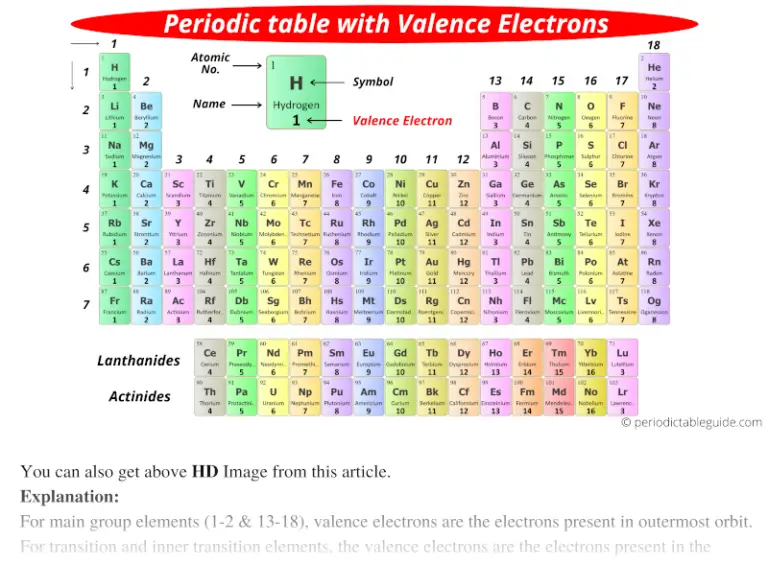 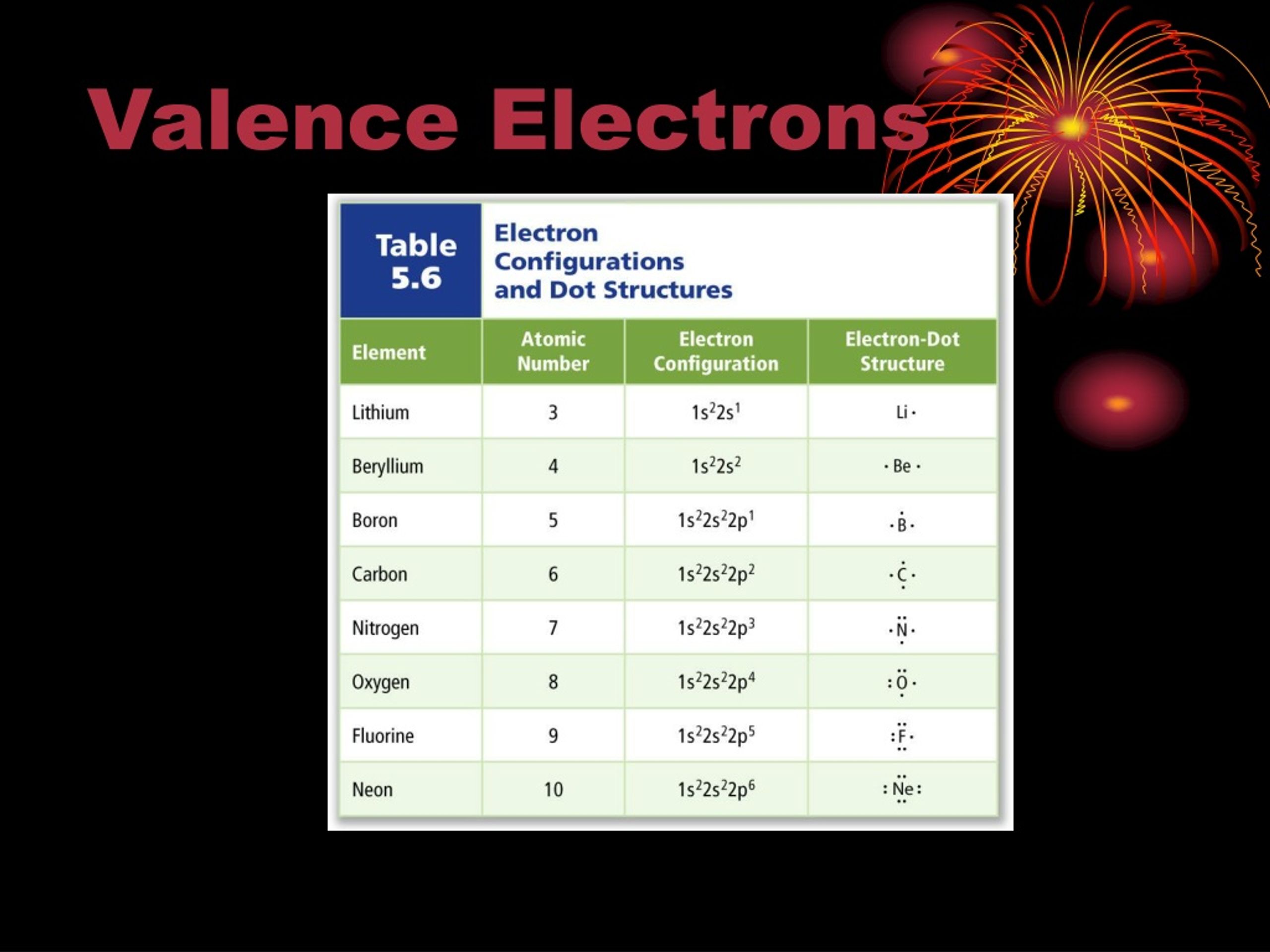
There are three degenerate 2 p orbitals ( m l = −1, 0, +1) and the electron can occupy any one of these p orbitals. Valence electrons of Elements (List) Here is the list of periodic table of elements with their valence electrons. Because any s subshell can contain only two electrons, the fifth electron must occupy the next energy level, which will be a 2 p orbital. The n = 1 shell is filled with two electrons and three electrons will occupy the n = 2 shell. So were going to ignore the other way to number the groups. E. When youre looking for the element, try to fill in from what youre given. The level of energy of an atoms valence electron(s) determines its period, (the horizontal lines). METALS The most reactive metals are those from Groups 1 and 2. Elements in a family (groups, the vertical bits) have the same number of valence electrons. Generally, elements in Groups 1, 2, and 13 to 17 tend to react to form a closed shell with a noble gas electron configuration ending in ns2np6. So one, two, three, four, five, six, seven, and eight. Elements whose atoms have the same number of valence electrons are grouped together in the Periodic Table. When we talk about the main groups, youre using the one through eight system for classifying groups. The fourth electron fills the remaining space in the 2 s orbital.Īn atom of boron (atomic number 5) contains five electrons. And so for this video, were only talking about the valence electrons for elements in the main groups. Recall, we can use the periodic table to rank the energy levels of various orbitals. The valence electrons, electrons in the outermost shell, are the determining factor for the unique chemistry of the element. 
Thus, the electron configuration and orbital diagram of lithium are:Īn atom of the alkaline earth metal beryllium, with an atomic number of 4, contains four protons in the nucleus and four electrons surrounding the nucleus. Many of the physical and chemical properties of elements can be correlated to their unique electron configurations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
